For families navigating an ultra-rare genetic disease diagnosis, the idea that only one type of treatment can be pursued at a time is not just incorrect. It can be genuinely harmful. ASOs modulate RNA while gene therapies deliver new genetic material for durable effects, and these mechanisms are not in competition. When you understand what each tool actually does at the molecular level, the case for combining them becomes not just logical, but often medically compelling. This guide breaks down how, when, and why these two approaches can work together to give patients with rare diseases better options.
Table of Contents
- Understanding ASOs and gene therapy: How they work
- Why combine both approaches? The clinical rationale
- Who benefits most? Patient scenarios for combination therapy
- Limits, risks, and what integration really means
- Future directions: Immune modulation and combination innovation
- A nuanced view: What most guides miss about combining ASOs and gene therapy
- How RareLabs can support your rare disease journey
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Layered mechanisms | Combining ASOs and gene therapy targets different biological levels for greater flexibility and effect. |
| Personalized treatment | The right combination depends on patient genetics, disease stage, and treatment goals. |
| Timing and safety | ASOs allow programmable, often reversible effects, while gene therapy offers lasting results but with possible immune risks. |
| No one-size-fits-all | Careful assessment and integration, not simple add-on therapy, is key for effective rare disease care. |
| Innovation ahead | Immune modulation and tailored combinations are rapidly advancing, offering new hope for rare conditions. |
Understanding ASOs and gene therapy: How they work
Now that we've set the stage, let's clarify what makes ASOs and gene therapies different but highly complementary.
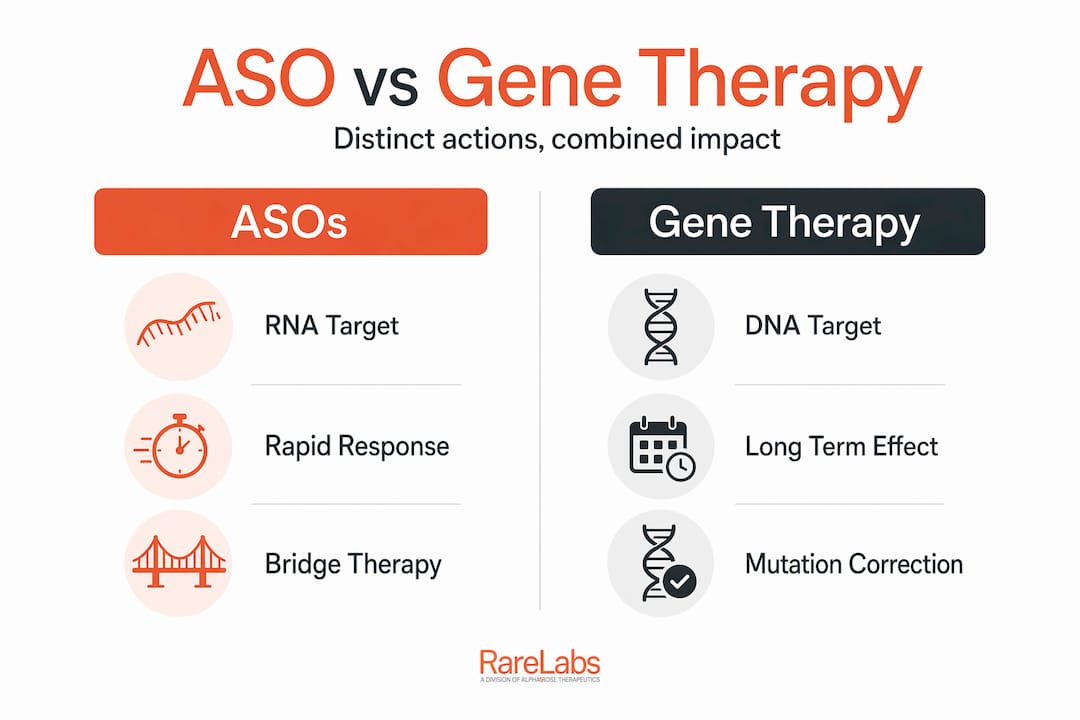
Antisense oligonucleotides, commonly called ASOs, are short synthetic strands of nucleic acid designed to bind to a specific RNA target inside cells. Once bound, they can do one of several things: trigger degradation of a problem RNA, correct faulty splicing so a gene produces the right protein, or block a toxic transcript from being translated at all. What makes ASOs particularly valuable is their precision and their programmability. Scientists can redesign an ASO relatively quickly if a patient's specific genetic variant requires a different target.
Gene therapy, by contrast, operates at the DNA level. The most established approach uses adeno-associated virus (AAV) vectors to deliver a functional copy of a gene directly into a patient's cells. Once delivered, the goal is long-lasting or even permanent correction. Some gene therapy strategies also use viral vectors to introduce gene editing tools like CRISPR directly. The appeal is obvious: one treatment that, in theory, fixes the underlying problem for years.
ASOs act at the RNA level, gene therapies at the DNA level; each offers unique strengths and limitations. Exploring gene therapy approaches and genetic therapies for rare diseases side by side makes the contrast sharper.

| Feature | ASO therapy | Gene therapy |
|---|---|---|
| Mechanism | RNA modulation | DNA delivery or editing |
| Duration of effect | Weeks to months (reversible) | Years (often long-lasting) |
| Reversibility | Yes, effect fades when dosing stops | Largely irreversible |
| Delivery route | Injection, intrathecal | IV, intrathecal, direct organ |
| Speed to customize | Weeks to months | Years |
| Key risk | Off-target effects, kidney toxicity | Immune response, insertional risks |
Key things to keep in mind when comparing them:
- ASOs can be custom-designed for a specific patient's variant far faster than a gene therapy can be manufactured and approved
- Gene therapies generally require only one or a few administrations, reducing long-term treatment burden
- ASOs require repeat dosing to maintain effect, which is both a limitation and a safety feature
- Gene therapy access is limited by manufacturing complexity and immunological eligibility
Pro Tip: For some rare mutations, picking the right combination depends on variant biology and tissue access. A patient with a CNS disease and a toxic gain-of-function mutation may benefit from an ASO to suppress the bad protein first, while a broader gene delivery strategy is prepared.
Why combine both approaches? The clinical rationale
Understanding the unique features of each approach leads to key reasons why clinicians may use both simultaneously or in sequence.
This is where the real conversation begins for families and physicians. Combining ASOs and gene therapy is not about throwing everything at a disease and hoping something sticks. It is a deliberate, strategic choice, and there are specific clinical rationales that support it.
-
Temporizing while gene therapy is developed. Custom gene therapies for ultra-rare diseases can take three to seven years to reach a patient after identification of the target. An ASO can be designed in months and used to stabilize a patient, slow progression, or suppress a toxic protein while the more durable gene therapy is prepared. This bridge strategy has real-world precedent.
-
Fine-tuning or reversing effects. Because ASO effects fade when dosing stops, they can serve as an adjustable dial. If a patient shows unexpected effects from modulating a gene product, dosing can be paused. Gene therapy lacks this property almost entirely once delivered.
-
Addressing immune response barriers. One of the most underappreciated challenges in gene therapy is the patient's immune system. Pre-existing immunity to AAV vectors, or an immune reaction triggered by the therapy, can dramatically reduce durability and safety. An ASO-based bridging approach can keep a patient stable while immune modulation protocols are designed and tested.
"ASO effects are reversible and programmable, while gene therapy may have limits due to immune responses." This programmability is not a secondary feature. It is clinically essential when you are dealing with a disease where biological variability between patients is extreme and where a wrong guess can have serious consequences.
This combination strategy addresses timing and risk tradeoffs that neither therapy alone handles well. A thorough review of gene therapy screening options and understanding gene therapy risks and expectations are essential steps before any combination plan is considered. The Nature review of combination approaches offers important scientific context for clinicians and informed families alike.
Who benefits most? Patient scenarios for combination therapy
With the clinical reasons clear, let's see what this means for patients and families facing tough choices.
Not every rare disease patient is a candidate for combined ASO and gene therapy. The decision depends heavily on the type of mutation, the disease's mechanism, the tissues involved, and the patient's current clinical status. Here is a practical breakdown of scenarios where combination strategies are most relevant.
| Patient scenario | Why combination makes sense |
|---|---|
| Gain-of-function mutation | ASO suppresses the toxic gene product; gene therapy later restores function |
| Dominant-negative mutation | Allele-selective ASO can reduce harmful allele while gene therapy prepares a functional copy |
| CNS disease with delivery challenges | Intrathecal ASO provides neurological access quickly; gene therapy targets broader correction |
| Loss-of-function, gene therapy under development | ASO stabilizes symptoms or modulates related pathways as a bridge |
| Immune-compromised patient | ASO buys time while immune eligibility for gene therapy is established |
Situations where families and clinicians should seriously consider a combined approach:
- The patient has a progressive disease and cannot wait years for a gene therapy to be developed
- The specific mutation causes a toxic protein that can be reduced by an ASO immediately
- Prior gene therapy attempts have been limited by immune responses
- The disease affects the CNS, where tailored ASOs provide individualized routes while gene therapy is developed
- The treating team needs reversibility as a safety net during early intervention
Allele-selective ASOs can reduce dominant-negative effects and serve as a genuine bridge until gene therapy becomes feasible. This matters enormously for conditions where only one copy of a gene is faulty and the goal is to silence just that copy without disrupting the healthy one. Understanding personalized rare disease treatments and identifying therapeutic targets for rare diseases are key steps in figuring out whether combination therapy is even possible for a specific patient.
Pro Tip: For dominant-negative mutations, always check whether an allele-selective ASO is possible before committing to other approaches. Reducing the toxic allele's expression can simplify the gene therapy design and reduce the burden of both treatments.
Limits, risks, and what integration really means
It's just as important to understand what this combination doesn't mean, and where families and clinicians should exercise caution.
The phrase "combination therapy" can sound like a simple upgrade, but in reality it introduces its own set of challenges. Integration of ASOs and gene therapy is not about taking two good things and adding them together. It requires careful, individualized planning based on the patient's biology, the disease's specific mechanism, and the timing of each intervention.
Real integration challenges that clinicians and families need to understand:
- Off-target toxicity from ASOs. Even well-designed ASOs can affect unintended RNA targets, particularly in kidney and liver tissues. This requires monitoring and sometimes dose adjustment.
- Repeated dosing requirements. ASOs typically require injections every four to twelve weeks. For intrathecal delivery (directly into spinal fluid for CNS diseases), this adds procedural burden and risk over time.
- Immune response to gene therapy vectors. Pre-existing antibodies to AAV, or an immune activation triggered by the vector, can limit the therapy's effectiveness or even cause serious side effects. Combining therapies amplifies the complexity of managing this.
- Defining the right patient group. Not all patients with the same diagnosis will benefit equally. The mutation type, disease stage, tissue involvement, and existing treatments all affect the risk-benefit calculation.
"Properly combining ASOs and gene therapy requires integration based on patient, tissue, variant, and timing — not simply adding one therapy on top of another." The design, delivery, and toxicity limitations of ASOs mean that every combined strategy must be built from the ground up for the individual patient, not copied from a general protocol.
A comprehensive gene therapy evaluation is essential before any combination approach is finalized. Clinicians and families should also review contrasting perspectives on what "combination" actually entails in practice. The honest message is that no combination strategy is inherently safer or better. It is only better when it is better for this patient, at this moment, with these options.
Future directions: Immune modulation and combination innovation
Having understood the current state, let's glimpse where research and clinical innovation are headed.
One of the most exciting frontiers in rare disease treatment is the integration of immune modulation into combination therapy strategies. A major reason gene therapies lose effectiveness over time is immune clearance. The body recognizes the viral vector or the introduced protein and mounts a defense. Researchers are now exploring ways to actively manage this immune response as part of the treatment plan, not just as a side effect to be managed.
Emerging innovations and research directions worth following:
-
PD-L1 and PD-L2 co-delivery with AAV vectors. These immune checkpoint ligands are being co-delivered with AAV vectors to suppress immune activation and prolong gene therapy expression. Early mouse model data from 2026 studies show meaningful improvements in durability when this strategy is used alongside targeted gene delivery.
-
Allele-selective ASO targeting. Increasingly, researchers are designing ASOs that can distinguish between the healthy and mutated copies of a gene at the single-nucleotide level. This precision opens the door to suppressing toxic proteins without affecting normal gene function, which makes subsequent gene therapy safer and simpler.
-
Combinatorial screening platforms. Labs are now using patient-derived iPSC (induced pluripotent stem cell) models to test ASO and gene therapy combinations in parallel, identifying which sequencing and dosing strategies produce the best outcomes before anything reaches a patient.
-
Exon skipping combined with gene augmentation. In diseases where a mutation causes a protein to be truncated or absent, an ASO that promotes exon skipping can produce a shorter but partially functional protein. This can reduce disease severity enough to extend the window for gene augmentation therapy.
Good disease modeling for rare disease therapies is at the center of all of this innovation. Without accurate models of what the disease actually does in a patient's own cells, combination strategies are educated guesses. With them, they become testable hypotheses.
A nuanced view: What most guides miss about combining ASOs and gene therapy
Most guides on this topic focus on mechanism. They explain what ASOs do, what gene therapy does, and why the mechanisms are theoretically compatible. What they rarely address is the harder reality: for most families, the decision about combining these approaches is not primarily a scientific one. It is shaped by access, timing, geography, clinical infrastructure, and the specific expertise available at a given center.
A family whose child has an ultra-rare CNS disorder may not be able to access an ASO program and a gene therapy program at the same institution. They may be navigating insurance barriers, travel requirements, and an exhausted support system at the same time. Understanding the science is necessary. But assuming that the best theoretical combination is always achievable in practice is where guides like this one often fall short.
Our perspective at RareLabs, shaped by working with families facing exactly these conditions, is that the goal is not to find the perfect combination. It is to find the best-informed choice available today that preserves the most options for tomorrow. That means individualized evaluation, not protocols. It means asking which ASO is accessible now, which gene therapy programs are enrolling or planning to enroll, and what the realistic timeline looks like. Gene editing and hope are real, but they are most valuable when grounded in an honest, specific assessment of what each patient can actually access and tolerate.
Even as immune modulation and combinatorial screening advance, the hard conversations about benefit and risk do not go away. They become more nuanced. That is a good thing for science. For families, it requires a guide who understands both the biology and the human stakes.
How RareLabs can support your rare disease journey
Navigating ASO and gene therapy options, separately or in combination, is one of the most complex challenges in rare disease medicine today. Families should not have to piece this together from scattered research papers alone.

RareLabs maintains a continuously updated knowledge center for rare disease research that covers combination strategies, emerging ASO developments, gene therapy trial access, and immune modulation research. More importantly, RareLabs builds personalized disease models from a patient's own cells, making it possible to test which approaches are most likely to work before committing to a clinical path. If you are a family, physician, or foundation trying to move from information to action, RareLabs is ready to help you find the most informed route forward for your specific situation.
Frequently asked questions
Are ASO and gene therapy ever given together at the same time?
Yes, they can be given sequentially or together in special cases to maximize benefit or manage risks, but this depends on the specific disease, mutation, and clinical context. Timing and benefit-risk tradeoffs are central to this decision.
Is combination therapy safer than just gene therapy alone?
Combination therapy is sometimes safer, but it also introduces new risks. Each strategy has a different risk profile, and combining them can create new challenges that must be carefully managed for each individual patient.
How do doctors decide which patients are suited for combination approaches?
Doctors review the specific genetic mutation, disease type, patient history, and the realistic benefit versus risk when considering a combination. Consensus guidance on eligibility for ASO and gene therapy combinations is still evolving, making individualized evaluation essential.
Will insurance cover both treatments?
Coverage varies widely depending on the payer, the country, and whether each treatment has regulatory approval. Both therapies typically require detailed medical necessity documentation, and families often need advocacy support to navigate approval processes.
Are there examples where combining these approaches actually improved long-term outcomes?
Emerging research shows that immune modulation strategies like PD-L1/2 co-delivery can improve gene therapy durability in preclinical models. Results in individual patients depend heavily on disease type, mutation, and how the therapies are sequenced and monitored over time.
