When families first encounter the phrase "ex vivo gene therapy," it often arrives buried in a clinical consult or a research paper, sitting uncomfortably close to "in vivo gene therapy" as if the two terms are interchangeable. They are not. The distinction matters enormously, because ex vivo gene therapy removes a patient's cells, modifies them outside the body, and returns them, while in vivo therapy delivers genetic material directly inside the patient. For families navigating ultra-rare and undiagnosed genetic diseases, understanding which approach fits your situation can shape which clinical trials you qualify for and what conversations to have with your care team.
Table of Contents
- What is ex vivo gene therapy?
- Step-by-step: How does ex vivo gene therapy work?
- Key technologies: Vectors and gene-editing tools
- Why choose ex vivo? Advantages for ultra-rare diseases
- Ex vivo vs. in vivo gene therapy: How do they differ?
- Real-world examples and regulatory progress
- Our take: What most guides miss about ex vivo gene therapy
- Next steps: Finding gene therapy options for your rare disease
- Frequently asked questions about ex vivo gene therapy
Key Takeaways
| Point | Details |
|---|---|
| Ex vivo explained | Ex vivo gene therapy removes, modifies, and reinfuses a patient's cells to treat genetic diseases. |
| Ultra-rare disease focus | This approach is especially important when traditional treatments are unavailable or ineffective. |
| Personalized treatment | Therapies can be custom-made for each patient, especially with autologous cell products. |
| Safety checks | Rigorous testing of modified cells before use increases safety for patients. |
| Real-world approvals | FDA-approved ex vivo therapies now exist for some ultra-rare genetic diseases. |
What is ex vivo gene therapy?
At its core, ex vivo gene therapy removes cells, genetically modifies them outside the body, and returns them to the patient. The Latin phrase "ex vivo" simply means "outside the living." Think of it like taking a faulty component out of a machine, repairing it on a workbench with the best tools available, and then reinstalling it. Nothing is rushed. Every step happens in a controlled environment where scientists can verify the repair worked before anything goes back into the patient.
The cells used in ex vivo therapy are almost always ones the body produces in large numbers and depends on heavily. Blood-forming cells, called hematopoietic stem and progenitor cells, are the most common. Immune cells like T cells and skin cells are also well-established targets. These cell types are practical choices because they can be collected without major surgery and can engraft, or settle and multiply, once returned to the patient.
"Ex vivo gene therapy is not just a technical workaround. It is a deliberate choice to maximize control, safety testing, and product quality before a patient ever receives modified cells."
The contrast with in vivo therapy is worth stating plainly. In vivo gene therapy delivers its genetic payload directly into the patient's body, often through an intravenous injection or a local injection to a target tissue. There is no cell removal, no workbench repair, and no pre-infusion quality check on each treated cell. Both approaches have merit, but they serve different diseases and different circumstances. Reviewing the must-know gene therapy approaches used for ultra-rare diseases provides helpful context for how ex vivo fits into the broader landscape.
Key features of ex vivo gene therapy:
- Cells are collected from the patient (autologous) or a matched donor (allogeneic)
- Genetic modification happens in a specialized lab setting under controlled conditions
- The modified cell product is tested for safety and effectiveness before reinfusion
- The patient typically undergoes a conditioning regimen to prepare the body to receive modified cells
- The therapeutic effect is designed to be long-lasting or permanent
Pro Tip: When reviewing a potential ex vivo trial for your loved one, always ask the clinical team what percentage of modified cells are expected to engraft and persist. This number directly connects to how durable the treatment effect may be.
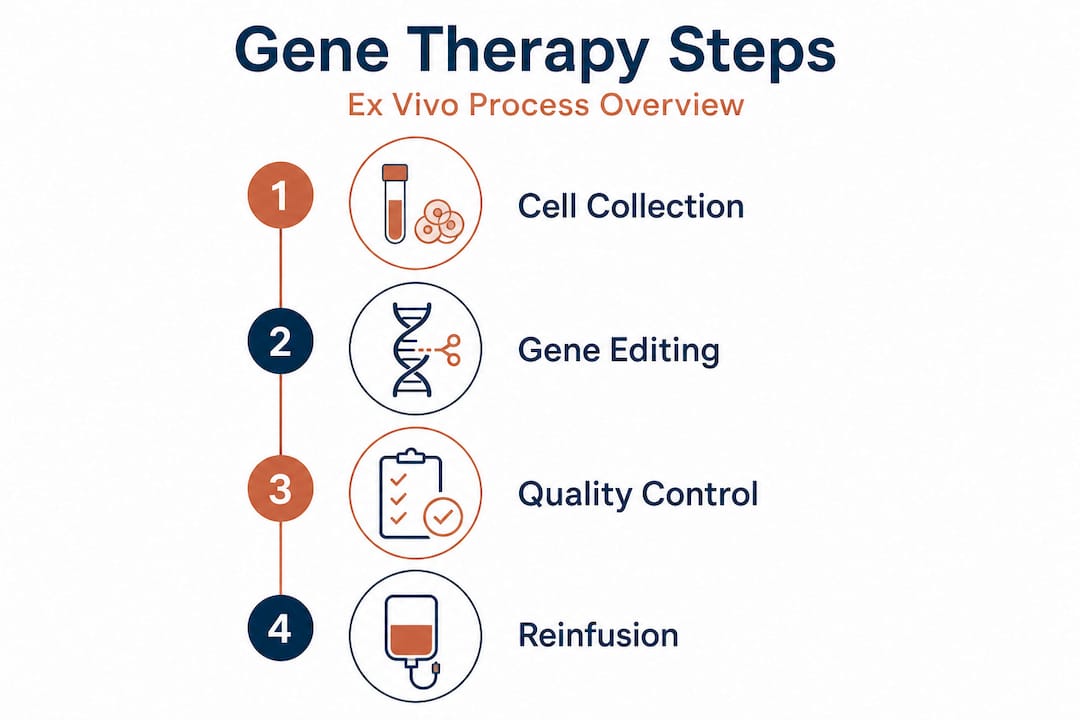
Step-by-step: How does ex vivo gene therapy work?
Understanding the sequence of steps removes a lot of the anxiety families feel when first considering this treatment path. The typical ex vivo process follows four major stages: harvesting target cells, modifying them with a vector or gene-editing system, performing quality controls on the cell product, and reinfusing after conditioning. Here is what each step actually involves.
-
Cell collection. Blood-forming stem cells are most often collected via apheresis, a process where blood is drawn, stem cells are separated, and the rest is returned. A growth factor called G-CSF is usually given beforehand to push stem cells out of the bone marrow and into the bloodstream. Skin cells and other tissue-specific cells require a small biopsy.
-
Genetic modification. Collected cells travel to a specialized manufacturing facility. There, scientists introduce the corrected or replacement gene using a viral vector, most often a lentiviral vector, or use a genome editing tool like CRISPR/Cas9 to correct the faulty sequence directly in the DNA.
-
Quality control. This is where ex vivo truly earns its safety advantage. The modified cell product is tested for gene insertion efficiency, cell viability, sterility, and the absence of unintended genetic changes. Products that do not meet strict specifications are not used.
-
Conditioning and reinfusion. Before reinfusion, the patient often undergoes a conditioning regimen using chemotherapy or targeted antibody therapy. This clears space in the bone marrow for modified cells to engraft. The modified cells are then infused, usually through a standard IV, much like a blood transfusion.
| Process stage | Typical duration | Key action |
|---|---|---|
| Cell collection | 1 to 2 days | Apheresis or biopsy |
| Manufacturing and modification | 2 to 6 weeks | Vector transduction or gene editing |
| Quality control testing | Concurrent with manufacturing | Viability, safety, and potency checks |
| Conditioning regimen | 3 to 10 days | Chemotherapy or immunodepletion |
| Reinfusion and monitoring | Ongoing months to years | Engraftment and long-term follow-up |
For ultra-rare diseases, this timeline can stretch longer if a custom vector or novel editing construct must be manufactured specifically for the patient's genetic variant. The step-by-step gene therapy screening process used in rare disease trials explains how this evaluation unfolds in practice.
Pro Tip: Ask the trial sponsor or treating center for their specific manufacturing timeline upfront. A 6-week manufacturing window affects travel, school, work, and caregiver planning in ways that generic timelines do not capture.
Key technologies: Vectors and gene-editing tools
The tools used to modify cells in ex vivo therapy are advancing rapidly, and knowing the basics helps you ask better questions. Commonly used approaches include viral vectors such as lentiviral and retroviral vectors, along with genome editing technologies such as CRISPR/Cas9. Each has a different mechanism, risk profile, and track record.

| Technology | Mechanism | Primary advantage | Key consideration |
|---|---|---|---|
| Lentiviral vector | Integrates a new gene copy into the genome | Proven in blood diseases, durable expression | Random integration site requires monitoring |
| Retroviral vector | Similar to lentiviral, targets dividing cells | Strong history in early gene therapy trials | Risk of insertional mutagenesis is lower with modern designs |
| CRISPR/Cas9 | Cuts and corrects the specific faulty DNA sequence | Highly precise, targets the root mutation | Delivery and off-target editing still being refined |
| Base and prime editing | Newer forms of CRISPR that change single DNA letters without cutting | Minimal off-target risk | Earlier stage of clinical development |
Because cells are modified and tested before they ever enter the patient's body, the ex vivo approach gives scientists the ability to screen the product for off-target edits or incomplete correction before infusion. This quality gate is not available in in vivo therapy, where the vector or editing tool acts inside the patient's tissues in real time.
Why this matters for ultra-rare diseases specifically:
- Many ultra-rare diseases are caused by a single-gene mutation, making them ideal targets for precise gene correction
- The relatively small patient populations mean custom vectors can be designed for a specific mutation rather than a common variant
- CRISPR-based therapeutic targets for rare diseases are expanding quickly, with dozens of investigational programs now in early clinical phases
- Pre-infusion testing means families can have documented proof that the gene correction was achieved before treatment proceeds
Why choose ex vivo? Advantages for ultra-rare diseases
For many ultra-rare genetic conditions, ex vivo gene therapy is the preferred route not because it is the newest technology, but because it fits the biology of the disease. Ex vivo therapy is favored where target cells are accessible, allowing manipulation in a controlled setting and rigorous pre-infusion testing. This is a fundamentally different kind of confidence than any in vivo approach can currently offer.
Blood disorders like sickle cell disease and beta-thalassemia, rare immune deficiencies, and certain skin disorders are all conditions where the target cells can be cleanly collected and returned. This accessibility is why ex vivo approaches have regulatory precedents using autologous gene-modified cell products for ultra-rare genetic diseases, a fact that matters when families are assessing whether a path to treatment even exists.
"The ability to verify that the gene was successfully corrected in your specific cells, before those cells go back into your body, is one of the most meaningful safety advantages modern medicine can offer a patient with an ultra-rare disease."
Specific advantages worth discussing with your clinical team:
- Pre-infusion safety verification: Each cell product batch is tested before use, catching manufacturing failures before they reach the patient
- Personalized to your mutation: Autologous designs use the patient's own cells, reducing rejection risk and enabling mutation-specific correction
- Potentially one-time treatment: Engrafted modified stem cells can self-renew, meaning the therapy may last a lifetime
- Regulatory framework exists: Approved therapies demonstrate the pathway is viable, not purely experimental
Exploring the full range of personalized rare disease therapies and genetic therapy options can help families see where ex vivo fits among the available approaches.
Pro Tip: If your disease involves a blood or immune cell lineage, ask your physician specifically about ex vivo options even if no approved therapy exists yet. Clinical trials and named patient programs sometimes operate ahead of formal approval.
Ex vivo vs. in vivo gene therapy: How do they differ?
Knowing the differences between ex vivo and in vivo approaches helps families and clinicians have more productive conversations about which path makes sense. The choice depends on accessibility of target cells, vector-delivery constraints, and patient-specific factors, not simply which technology is more advanced.
| Feature | Ex vivo gene therapy | In vivo gene therapy |
|---|---|---|
| Cell modification site | Outside the body, in a lab | Inside the patient's body |
| Target cell accessibility | Requires removable cell types | Can reach deep tissues |
| Pre-treatment testing | Extensive, on each product batch | Limited once delivered |
| Logistics | Complex manufacturing required | Simpler delivery process |
| Risk of immune response | Lower (autologous cells) | Higher (vector immune response possible) |
| Current approvals for ultra-rare | Growing list of precedents | Also active, different disease set |
The key factors for your specific situation:
- Disease biology. Does the disease affect cells that can be safely removed and returned? Blood and skin cells: yes. Brain neurons: not currently.
- Patient condition. Can the patient tolerate a conditioning regimen? This is often the most critical logistics question families face.
- Regulatory landscape. Are ex vivo trials or approved products available for your specific disease and mutation?
Reviewing a thorough gene therapy evaluation guide is a useful next step once you know your disease's target cell type.
Real-world examples and regulatory progress
Abstract science becomes real when you see actual approved products. FDA-approved examples include SKYSONA (elivaldogene autotemcel), an autologous hematopoietic stem cell gene therapy for cerebral adrenoleukodystrophy, and ZEVASKYN (prademagene zamikeracel), an autologous skin cell gene therapy for recessive dystrophic epidermolysis bullosa, a devastating rare skin disorder. Both represent diseases once considered untreatable with any targeted intervention.
| Approved therapy | Disease | Cell type used | Regulatory status |
|---|---|---|---|
| SKYSONA | Cerebral adrenoleukodystrophy | Hematopoietic stem cells | FDA approved |
| ZEVASKYN | Recessive dystrophic epidermolysis bullosa | Skin keratinocytes | FDA approved |
| CASGEVY | Sickle cell disease, beta-thalassemia | Hematopoietic stem cells | FDA approved |
Beyond approved products, the NINDS URGenT initiative explicitly identifies ex vivo gene targeting and cell therapies as priority approaches for ultra-rare neurological diseases. This is a meaningful signal that government funding and infrastructure are moving to support the next generation of treatments for diseases that previously had no research pathway.
What these approvals mean for families:
- The regulatory framework for ex vivo autologous therapies is established and growing
- Diseases that affect accessible cell types have the clearest path to approved therapy
- Even in the absence of an approved product, existing precedents support the design of new trials
The guide to finding rare disease treatments walks through how to use existing approval data to identify open trials and access programs relevant to your disease.
Our take: What most guides miss about ex vivo gene therapy
Most explanations of ex vivo gene therapy focus entirely on the science. They explain vectors, describe CRISPR mechanisms, and list approved products. What they rarely address is the human decision layer underneath all of it.
The ex vivo versus in vivo debate is not actually about which technology is superior. It is about fit. The right therapy is the one designed for the biology of your specific disease, the cells that carry the mutation, and the realities of your family's situation. A therapy that requires 6 weeks of manufacturing, a conditioning regimen, and a 3-month hospital stay near a specialized center is not the right choice for every family, even if the science is flawless.
What families actually need from their medical team is not a lecture on lentiviral vectors. They need clear answers to four questions. Can my loved one tolerate conditioning? How confident are we that engraftment will occur? What does long-term monitoring look like? And is there a real path to access, whether through a trial, an expanded access program, or an approved product?
We have seen families spend months researching gene therapy biology when what would have actually moved the needle was a single direct conversation with a metabolic disease specialist who knew the current trial landscape. Science literacy matters. But the right question to ask your clinical team is not "how does CRISPR work?" It is "what quality control data will exist on my child's specific cell product before it goes back into their body?"
Understanding gene therapy expectations and risks from a family perspective bridges this gap between technical knowledge and actionable decision-making.
Next steps: Finding gene therapy options for your rare disease
If this article has helped clarify what ex vivo gene therapy is and whether it might apply to your situation, the next step is finding out what is actually available right now for your specific disease and mutation.

At RareLabs, we build patient-specific disease models from your own cells and run parallel screens across thousands of FDA-approved drugs, custom ASOs, and gene therapy options to identify viable paths forward. Our process is designed specifically for ultra-rare and undiagnosed diseases where no approved treatment exists. You can search our rare disease knowledge base to find disease-specific information and current research, or visit our rare disease treatment search platform to explore what personalized modeling could uncover for your condition. Families and clinicians working on the most urgent cases get a team that understands the science and the urgency equally.
Frequently asked questions about ex vivo gene therapy
What types of diseases can ex vivo gene therapy treat?
Ex vivo gene therapy is favored for diseases where relevant target cells are accessible, most often blood, immune, or skin cell lineages. Diseases affecting deep tissues or organs that cannot be safely biopsied are better served by in vivo approaches.
Is ex vivo gene therapy safe?
Safety is significantly enhanced because rigorous quality control is performed on the modified cell product before it is ever given to the patient. This pre-infusion testing catches manufacturing problems, incomplete gene correction, and contamination issues before treatment.
How is ex vivo gene therapy different from in vivo gene therapy?
Ex vivo gene therapy modifies cells outside the body, while in vivo therapy delivers new genes directly inside the patient without removing cells first. The ex vivo approach allows for pre-infusion testing; in vivo therapy does not.
Are there FDA-approved ex vivo gene therapies for ultra-rare diseases?
Yes. FDA-approved therapies including SKYSONA for cerebral adrenoleukodystrophy and ZEVASKYN for recessive dystrophic epidermolysis bullosa demonstrate that ex vivo gene therapy has a real and growing regulatory track record for ultra-rare genetic conditions.
