For families navigating a rare or undiagnosed genetic disease, the word "untreatable" can feel like a final answer. It isn't. Gene therapy has reshaped what's possible, moving from theoretical science to approved treatments for conditions that once had none. According to the FDA, gene therapy is a treatment approach that uses genetic material, DNA or RNA, to treat or prevent disease by changing how cells produce proteins or by modifying genetic instructions at the source. This guide walks through how gene therapy works, what approaches exist, how delivery methods differ, what risks demand attention, and what practical steps patients, families, and care teams can take right now.
Table of Contents
- What is gene therapy and how does it work?
- Gene therapy mechanisms: Approaches and options
- How gene therapy is delivered: Comparing in vivo and ex vivo
- Safety and monitoring in gene therapy: What patients and families need to know
- The real world of gene therapy: What patients, families, and professionals should know
- Find gene therapy solutions and support for rare diseases
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Gene therapy offers hope | Gene therapy provides new possibilities for treating rare and undiagnosed genetic diseases. |
| Multiple approaches exist | Gene therapy uses replacement, addition, silencing, or genome editing tailored to the disease. |
| Delivery methods matter | Choosing between in vivo and ex vivo affects effectiveness, safety, and patient experience. |
| Long-term monitoring is essential | Regular follow-up is needed to manage risks and support lasting benefits. |
| Expert guidance is key | Families and professionals should partner with specialists for personalized treatment and care. |
What is gene therapy and how does it work?
Gene therapy is not a single treatment. It's a category of medical interventions that share one core idea: fix the problem at the genetic level rather than managing symptoms downstream. That distinction matters enormously for rare diseases, where the root cause is often a single faulty gene, a deletion, duplication, or point mutation that disrupts how cells function.
At the most basic level, gene therapy delivers genetic material to cells using a vector, which acts as a delivery vehicle. Vectors are commonly engineered viruses, such as adeno-associated viruses (AAVs) or lentiviruses, that carry therapeutic genetic cargo into target cells without causing disease themselves. Non-viral vectors, including lipid nanoparticles, are increasingly used as well.
Delivery strategies fall into two categories: in vivo and ex vivo. In vivo means the vector is delivered directly into the patient, where it travels to target tissues. Ex vivo means cells are removed from the patient, modified in a laboratory setting, and then returned to the patient's body. Each approach carries specific advantages depending on the disease, the target tissue, and the patient's clinical situation.
There's a compelling reason to pay attention to how well gene therapy performs in rare diseases specifically. Success rates for gene therapy programs in rare diseases run at approximately 19%, compared to just 7.3% for overall drug development programs. That gap reflects the advantage of targeting a defined, known genetic cause, which is often the case in rare and ultra-rare conditions.
The major gene therapy approaches currently in use or under development include:
- Gene replacement: Delivering a working copy of a faulty or missing gene
- Gene addition: Adding a new gene to help cells compensate for disease
- Gene silencing: Reducing or switching off overactive or harmful gene expression
- Genome editing: Using tools like CRISPR-Cas9 to precisely repair mutations within the patient's own DNA
Each of these approaches addresses a different kind of genetic problem. Understanding which one applies to a specific diagnosis is the foundation of any serious treatment conversation.
Gene therapy mechanisms: Approaches and options
Now that we've established how gene therapy works, let's explore the different mechanisms and options for tailoring therapy to individual needs. One of the most important points for patients and physicians alike is that not every mechanism fits every disease. The choice of approach must match the specific genetic defect involved.
According to the FDA's consumer overview, gene therapy can be carried out by replacing a missing or faulty gene, adding a gene to help treat disease, turning off a disease-causing gene, or using genome editing technology to repair specific mutations. Each strategy has distinct clinical strengths and limitations.
Here's how the main approaches compare:
| Mechanism | Best suited for | Example conditions | Key advantage |
|---|---|---|---|
| Gene replacement | Loss-of-function mutations | Spinal muscular atrophy, hemophilia | Restores missing protein directly |
| Gene addition | Disease needing supplemental proteins | Certain immune deficiencies | Does not require removing existing gene |
| Gene silencing | Gain-of-function or toxic gene expression | Transthyretin amyloidosis | Stops harmful protein production |
| Genome editing | Precise single-gene mutations | Sickle cell disease, beta-thalassemia | Corrects the mutation at source |
When exploring genetic therapy options for ultra-rare diseases, the picture becomes even more nuanced. Many ultra-rare conditions have only a handful of known cases globally, meaning there is rarely an approved therapy waiting. Instead, families and care teams must evaluate experimental approaches, compassionate use programs, or research-backed interventions that may include antisense oligonucleotides (ASOs) alongside gene therapy strategies.
Matching therapeutic targets to the specific genetic architecture of the disease is not a formality. It is the single most important scientific decision in the entire process. A silencing strategy applied to a loss-of-function mutation would worsen the disease, not treat it. This is why genetic diagnosis, ideally including full genome or exome sequencing, must come before any therapy discussion.
Pro Tip: If you are a family or physician evaluating gene therapy options, map the mechanism to the molecular diagnosis first. Ask your geneticist or specialist specifically: is this a loss-of-function or gain-of-function mutation? Dominant or recessive? That single question narrows the viable therapeutic approaches significantly.
Key considerations when evaluating mechanisms include:
- Whether the gene is large or small (some genes are too large for certain viral vectors)
- Whether the target tissue is accessible by current delivery methods
- Whether the patient's immune system has prior exposure to the chosen vector
- Whether the disease requires a one-time correction or ongoing intervention
How gene therapy is delivered: Comparing in vivo and ex vivo
Understanding the mechanism is only half the story. How gene therapy is delivered can have a major impact on outcomes, safety profiles, patient logistics, and the complexity of care required.
Delivery strategies differ fundamentally between in vivo and ex vivo approaches. Each carries real advantages and real limitations that must be weighed carefully.

In vivo delivery involves administering the vector directly to the patient, most commonly through intravenous infusion, intrathecal injection (into the spinal fluid), or targeted organ delivery. The patient receives the therapy without cell extraction. This is generally less invasive and operationally simpler.
Ex vivo delivery is more complex but offers greater precision. Cells, often blood stem cells or T cells, are collected from the patient, modified in a specialized laboratory using the therapeutic gene, and then re-infused. The modification process can be more tightly controlled, and the cells can be verified before returning them to the patient.
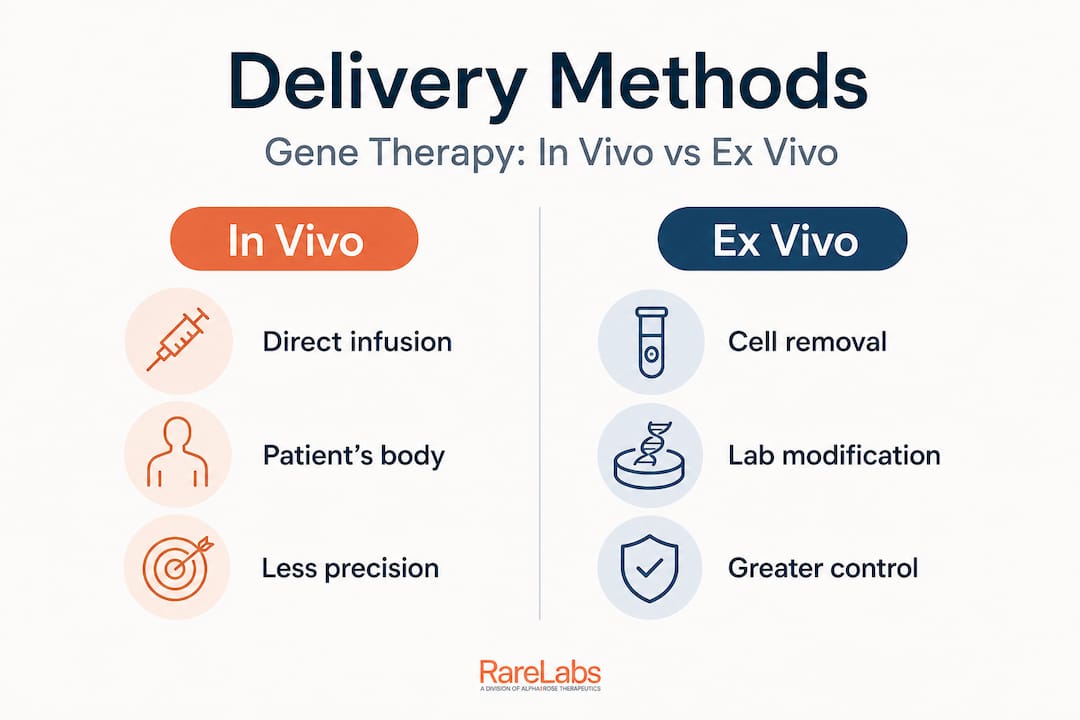
| Feature | In vivo | Ex vivo |
|---|---|---|
| Invasiveness | Lower (infusion or injection) | Higher (cell collection, conditioning, infusion) |
| Control over modification | Lower | Higher |
| Patient burden | Generally less | Requires hospitalization and preparation |
| Best suited for | Liver, CNS, muscle-targeted diseases | Hematopoietic and immune disorders |
| Immune risk | Immune response to vector | Immune response to modified cells |
Choosing between the two approaches follows a logical process:
- Identify the target tissue and whether it is accessible by direct delivery
- Evaluate the patient's immune status and prior vector exposure
- Assess whether the disease requires durable, permanent correction or staged intervention
- Review the patient's ability to tolerate conditioning regimens if ex vivo is planned
- Confirm center expertise in the chosen delivery approach
For families considering ex vivo gene therapy specifically, it is critical to understand that the process often requires the patient to undergo a conditioning regimen, similar to chemotherapy, to prepare the body to accept modified cells. This carries its own set of risks and demands careful patient selection.
"Not all gene therapy products behave the same—safety, conditioning, and immune response may differ substantially depending on vector, transgene, and patient-specific factors."
Research published in PMC confirms that safety and genotoxicity risk profiles vary based on the vector type and delivery approach used, underscoring why cookie-cutter comparisons between gene therapy products are clinically inappropriate. A thorough evaluation guide for rare disease gene therapies helps families and physicians work through these decisions systematically.
Safety and monitoring in gene therapy: What patients and families need to know
With delivery explained, safety and monitoring deserve special attention. These are the questions families ask most urgently, and they deserve honest, evidence-grounded answers rather than reassuring generalities.
The European Medicines Agency classifies gene therapies as advanced therapy medicinal products, recognizing that they work by inserting recombinant genes into the body to produce a therapeutic, prophylactic, or diagnostic effect. This classification carries strict regulatory requirements for manufacturing, clinical testing, and post-market surveillance.
The major safety considerations in gene therapy include:
- Immune response to the vector: The immune system may recognize the viral vector as foreign and mount a response, reducing efficacy or causing inflammation
- Immune response to the transgene product: The newly expressed protein may also trigger immune reactions, particularly if it's a protein the patient has never produced before
- Genotoxicity: With integrating vectors, there is a risk that the inserted gene could disrupt other genes, potentially leading to adverse long-term effects
- Off-target effects: Genome editing tools can sometimes cut DNA at unintended locations
- Delayed adverse events: Some risks do not appear immediately and may take months or years to manifest
Published safety data makes clear that safety and efficacy depend strongly on immune responses to vectors and transgene products, which can limit durability or create safety risks. This is not a reason to avoid gene therapy. It is a reason to approach it at centers with deep expertise and robust monitoring protocols.
Long-term follow-up is not optional. Because gene therapy can carry risks related to genotoxicity and delayed adverse events, monitoring after treatment is a formal expectation for advanced therapies, especially those using integrating vectors.
Pro Tip: Before enrolling in any gene therapy trial or program, families should ask the care team three specific questions: What vector is being used, and what is its known genotoxicity profile? What is the monitoring schedule and duration? How will immune responses be detected and managed? Centers that cannot answer these clearly are not ready to administer the therapy.
A dedicated parent guide to gene therapy risks provides a practical framework for families navigating these conversations. Understanding screening for gene therapy trials is equally important, since not every patient qualifies for every trial, and eligibility criteria vary widely based on age, disease stage, prior treatments, and immune status.
The real world of gene therapy: What patients, families, and professionals should know
Here's something most articles won't say plainly: gene therapy is not a single category of treatment you either get or don't get. It is a spectrum of approaches, mechanisms, delivery systems, and risk profiles, and the outcomes depend enormously on whether the right therapy was matched to the right patient with the right disease at the right stage.
We've seen families arrive having read that "gene therapy cured" a condition superficially similar to their child's diagnosis, only to find that the mechanism used, the delivery method, or the target tissue is completely different from what their disease requires. The success story is real. It just doesn't transfer automatically.
The most important lesson from working at the front of rare disease research is this: personalized care for rare diseases is not a marketing phrase. It is a scientific requirement. A therapy built for one genetic defect in one tissue can be ineffective or even harmful for a mutation in a similar gene. The specifics matter more than the category.
What most articles miss when covering gene therapy:
- Edge-case immune risks are under-discussed. Patients who have been previously exposed to a common AAV serotype may have pre-existing antibodies that neutralize the therapy before it works. Baseline immune screening is essential.
- Long-term monitoring is frequently underestimated. Regulatory bodies require years of follow-up for a reason. Families and physicians need infrastructure and commitment for this, not just the treatment itself.
- Therapy access is uneven. Approved therapies exist for a small number of rare diseases. For most ultra-rare conditions, the path involves research programs, clinical trials, or custom approaches built specifically for that disease.
Physicians and biopharma professionals need to hold two truths simultaneously: gene therapy has produced genuine, documented cures for conditions that previously offered only palliative options. And gene therapy for most rare diseases remains a work in progress, requiring patient-specific disease models, rigorous preclinical validation, and thoughtful clinical translation. The answer to "is gene therapy an option for my patient?" is almost always "it depends, and here's how to find out."
Find gene therapy solutions and support for rare diseases
Understanding the science is one thing. Knowing where to go next is another.

RareLabs builds patient-specific disease models from a patient's own cells using iPSC technology and CRISPR editing, then runs parallel treatment screens across FDA-approved drugs, custom ASOs, and gene therapy options to identify viable paths where none exist yet. If you're a family, physician, or biopharma partner working through a rare or undiagnosed genetic disease, explore the rare disease treatment knowledge hub for in-depth resources, or visit the RareLabs platform to learn how personalized disease modeling can accelerate the search for effective treatment options for your specific case.
Frequently asked questions
Is gene therapy safe for children with rare diseases?
Gene therapy safety depends on the delivery method, vector type, and patient immune response; long-term monitoring is required because risks like genotoxicity and delayed adverse events may not appear immediately after treatment.
How long does gene therapy last once given?
Durability varies by therapy and disease; some are designed for lasting correction while others may need repeat administration, since immune responses to vectors or transgene products can reduce how long the therapy remains effective.
Are all gene therapies the same?
No. Gene therapies use different mechanisms, including gene replacement, addition, silencing, and genome editing, each matched to a specific type of genetic defect and delivered through different methods.
What is the difference between in vivo and ex vivo gene therapy?
In vivo therapy is delivered directly into the patient's body, while ex vivo approaches involve removing cells, modifying them outside the body in a lab, and then returning them to the patient.
What monitoring is required after gene therapy?
Patients require regular follow-up for years after treatment; genotoxicity risks and delayed adverse events mean that structured long-term surveillance is a formal safety expectation for advanced gene therapies, particularly those using integrating vectors.
