Over 7,000 known diseases trace back to a single faulty gene. Not dozens of genes working together. Not environmental triggers stacked on top of complex biology. Just one gene, one mutation, and a cascade of consequences that can reshape a family's entire future. Many people assume all genetic diseases work the same way, requiring the same tools and carrying the same odds. That assumption leads to delayed diagnoses, missed carrier testing, and families left without a roadmap. The reality is that monogenic diseases are both more specific and more actionable than most people realize, and biotechnology is now making personalized treatment a genuine possibility.
Table of Contents
- What are monogenic diseases?
- Monogenic inheritance patterns: How these diseases are passed down
- Diagnosis and real-world impact: What patients and families should know
- Biotech breakthroughs: Personalized treatments for monogenic diseases
- A new perspective: Beyond the old boundaries of monogenic disease
- Finding support: Explore new treatment options with RareLabs
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Single-gene cause | A monogenic disease is caused by a mutation in just one gene, unlike more complex disorders. |
| Inheritance patterns vary | Monogenic diseases follow specific inheritance patterns, affecting risk for family members. |
| Diagnosis guides treatment | Genetic testing precisely identifies the disease, enabling more tailored care for patients. |
| Biotech is changing care | Innovative treatments like gene therapy and CRISPR are beginning to offer hope for some monogenic conditions. |
| Family planning impact | Understanding inheritance and genetic risks is vital for families and advocacy groups. |
What are monogenic diseases?
A monogenic disease is a health condition caused by a mutation in a single gene. That single change can disrupt a protein, silence a critical function, or trigger the production of something harmful. The result is a disease that follows predictable biological rules, which is both the challenge and the opportunity.
Most people are more familiar with polygenic or multifactorial conditions, like heart disease or type 2 diabetes, where risk comes from many genes interacting with lifestyle and environment. Monogenic diseases are different. They are driven by one gene event, which means the cause is often identifiable, the inheritance pattern is traceable, and the target for treatment is specific.
Here is a quick comparison to make the distinction clear:
| Feature | Monogenic disease | Polygenic/multifactorial disease |
|---|---|---|
| Cause | Single gene mutation | Multiple genes plus environment |
| Inheritance | Follows Mendelian patterns | Complex, less predictable |
| Diagnosis | Targeted genetic test | Broader risk scoring |
| Treatment target | Specific gene or protein | Multiple pathways |
| Examples | Cystic fibrosis, sickle cell | Type 2 diabetes, hypertension |
There are approximately 7,000 monogenic diseases identified to date, affecting tens of millions of people worldwide. Many are rare individually, but together they represent a massive unmet medical need.
Some of the most recognized examples include:
- Cystic fibrosis caused by mutations in the CFTR gene, affecting lung and digestive function
- Sickle cell disease caused by a single nucleotide change in the HBB gene, altering red blood cell shape
- Huntington's disease caused by an expanded repeat in the HTT gene, progressively damaging the nervous system
- Phenylketonuria (PKU) caused by mutations in PAH, leading to toxic amino acid buildup
- Duchenne muscular dystrophy caused by mutations in the DMD gene, affecting muscle protein production
For families navigating these diagnoses, knowing that a single gene is responsible is not just a scientific detail. It is the foundation for every decision that follows, from carrier testing to evaluating gene therapy treatments that target the exact mutation involved.
Monogenic inheritance patterns: How these diseases are passed down
After explaining the basics of what monogenic diseases are, it is important to look at how these disorders are inherited within families. Inheritance patterns determine who in a family is at risk, how likely a child is to be affected, and what genetic testing makes sense.
Monogenic diseases follow Mendelian patterns: autosomal dominant, autosomal recessive, X-linked dominant, and X-linked recessive. Each pattern carries different implications.
| Inheritance type | Risk to children | Carrier status | Example |
|---|---|---|---|
| Autosomal dominant | 50% per child | One copy causes disease | Marfan syndrome, Huntington's |
| Autosomal recessive | 25% if both parents carry | Two copies needed | Cystic fibrosis, PKU |
| X-linked recessive | 50% of sons affected | Daughters often carriers | Hemophilia, Duchenne MD |
| X-linked dominant | Variable | One copy on X causes disease | Fragile X syndrome |
Here is how to think through each one:
- Autosomal dominant means one mutated copy of the gene is enough to cause the disease. A parent with the condition has a 50% chance of passing it to each child, regardless of sex.
- Autosomal recessive means a child must inherit two mutated copies, one from each parent. Parents who each carry one copy are often completely healthy but face a 25% chance per pregnancy of having an affected child.
- X-linked recessive conditions affect males far more often because they carry only one X chromosome. Females with one affected copy are usually carriers without symptoms, but X-linked disorders like hemophilia can still cause mild symptoms in some female carriers.
- X-linked dominant conditions can affect both sexes, though severity often differs.
Understanding which pattern applies to your family changes everything. It tells you whether siblings are at risk, whether your children need testing, and whether a pregnancy warrants preimplantation genetic testing.
Pro Tip: If a monogenic disease runs in your family, ask your physician for a referral to a genetic counselor before starting a family. Carrier testing is often straightforward and can dramatically clarify your actual risk.
Diagnosis and real-world impact: What patients and families should know
With inheritance patterns clarified, the next concern for patients and advocates is how monogenic diseases are actually diagnosed and what the diagnosis truly means.
Genetic testing is the cornerstone. A targeted DNA test can identify the specific mutation responsible, confirming a diagnosis and enabling precise family planning. But the process is rarely a single step. Most families move through several stages:
- Clinical evaluation based on symptoms, family history, and physical findings
- Targeted gene panel or whole exome sequencing to identify the causative variant
- Confirmation testing in family members to establish carrier status or inheritance pattern
- Specialist referral to connect the genetic finding with disease management options
- Ongoing monitoring because many monogenic diseases evolve over time
One thing that surprises many families is that not everyone who carries a disease-causing mutation will develop the condition. This is called incomplete penetrance. A person can carry the exact same mutation as an affected relative and remain symptom-free, or develop a much milder form of the disease. Polygenic background modifies risk, age of onset, and symptom severity in ways that are still being studied. For example, roughly 24% of MODY (a monogenic form of diabetes) risk is explained by polygenic factors, not just the primary mutation.
"Not every mutation tells the whole story. The genetic background a person inherits alongside a primary variant can shift outcomes significantly, which is why personalized modeling matters so much."
Pro Tip: If your genetic test comes back positive but your symptoms do not match the expected picture, ask specifically about penetrance and expressivity. A specialist familiar with your condition can help you interpret what the result actually means for your health.
For families, the diagnosis is not just medical. It affects insurance decisions, family planning, career choices, and emotional wellbeing. Advocacy organizations and specialized biotech labs can help translate a genetic finding into a practical action plan.

Biotech breakthroughs: Personalized treatments for monogenic diseases
Understanding diagnosis leads many to wonder what can actually be done. This is where biotech is changing the field in ways that felt impossible just a decade ago.
Gene therapy is the most direct approach. Instead of managing symptoms, it targets the root cause: the gene itself. The main platforms in use today include:
| Technology | Mechanism | Status |
|---|---|---|
| CRISPR-Cas9 | Cuts and edits DNA at a specific site | FDA-approved (CASGEVY) |
| Base editing | Changes a single DNA letter without cutting | Clinical trials |
| AAV vectors | Delivers a working gene copy into cells | Multiple approvals |
| Antisense oligonucleotides (ASOs) | Modifies gene expression at RNA level | Multiple approvals |
The most striking recent example is CASGEVY, the first CRISPR-based therapy approved by the FDA. In clinical data, 96.7% of sickle cell patients treated with CASGEVY remained free of vaso-occlusive crises at 12 or more months. That is not symptom management. That is a functional cure for a disease that once meant a lifetime of pain episodes and hospitalizations.
Beyond CRISPR, gene editing platforms including AAV vectors are enabling one-time treatments for conditions like spinal muscular atrophy, hemophilia, and certain forms of inherited blindness. The pipeline for 2026 includes dozens of monogenic conditions moving through late-stage trials.
For families dealing with ultra-rare or undiagnosed conditions, certified gene therapy options are expanding rapidly. The key is connecting the specific mutation to the right platform, which requires patient-specific modeling rather than a one-size-fits-all approach.
Statistic to know: The global gene therapy market is projected to exceed $25 billion by 2030, driven almost entirely by monogenic disease applications.
A new perspective: Beyond the old boundaries of monogenic disease
With these exciting advances in view, it is vital to reconsider what "monogenic" really means in 2026. The traditional picture was clean: one rare variant, complete penetrance, clear inheritance. Either you had the mutation or you did not.
That picture is being redrawn. Emerging research shows that the boundary between monogenic and polygenic disease is a continuum, not a wall. Some common variants with strong effects behave more like monogenic risk factors. Some classic monogenic mutations cause wildly different outcomes depending on polygenic background. The old labels are becoming less useful for families trying to make real decisions.
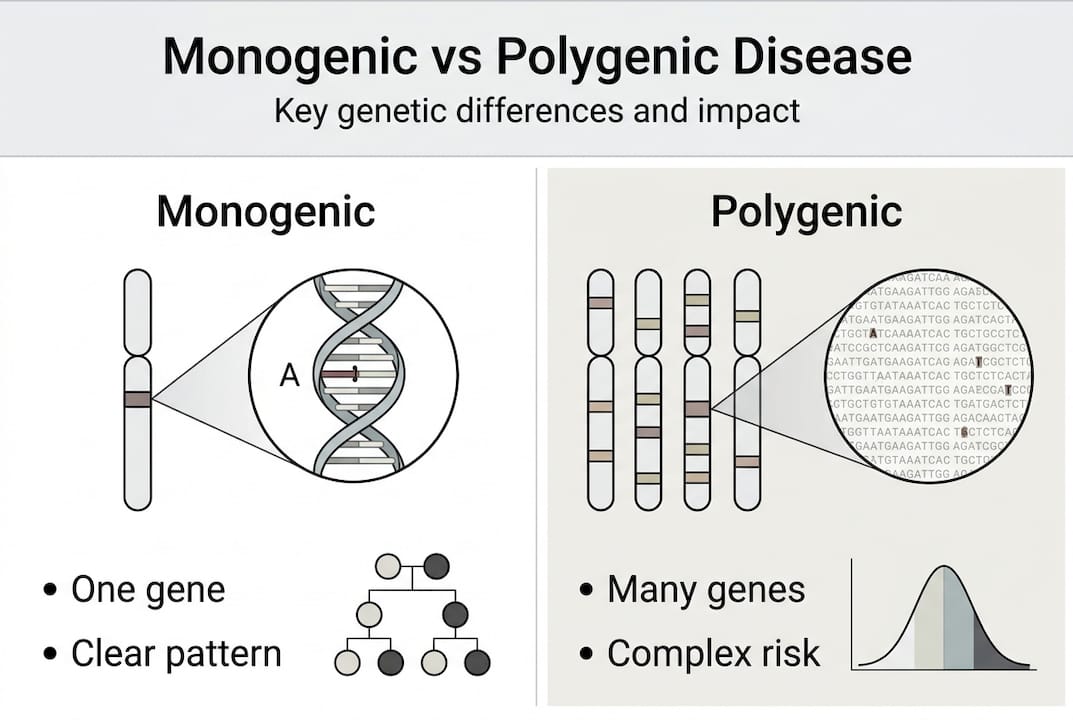
What this means practically is that a negative genetic test is not always reassuring, and a positive one is not always a sentence. Families who access genetic disease insights from specialized labs, rather than relying on general population statistics, get a far more accurate picture of their actual risk and options.
The most empowering shift is this: monogenic disease is no longer a category defined by what cannot be done. It is a category defined by a specific, targetable cause. That specificity is exactly what modern biotech was built to address.
Finding support: Explore new treatment options with RareLabs
For patients, families, and advocates ready to take the next step in the search for innovative solutions, here is how to begin.

At RareLabs, we build patient-specific disease models using iPSCs and CRISPR technology derived from your own cells. We then run parallel screens across thousands of FDA-approved drugs, custom ASOs, and RareLabs treatment search options to find what works for your exact mutation. Whether you are newly diagnosed, years into a rare disease journey, or advocating for someone you love, we offer a scientifically rigorous, transparent process designed to move fast when time matters most. Reach out to our team to learn what personalized treatment modeling could mean for your family.
Frequently asked questions
What is a monogenic disease in simple terms?
A monogenic disease is a health condition caused by a change in a single gene, like cystic fibrosis or sickle cell disease. One gene, one mutation, one root cause.
How do monogenic diseases differ from polygenic diseases?
Monogenic diseases are caused by mutations in a single gene, while polygenic diseases involve the combined effect of multiple genes. The distinction shapes risk assessment, diagnosis, and treatment strategy significantly.
Can monogenic diseases be cured or treated?
Some monogenic diseases can now be treated or even functionally cured using gene therapy. The CRISPR-based CASGEVY approval for sickle cell disease is the clearest example of this shift.
How are monogenic diseases diagnosed?
Diagnosis usually involves genetic testing to find the single gene mutation responsible. Genetic testing identifies the causative variant and guides both family planning and treatment decisions.
Are all monogenic diseases inherited in the same way?
No. They can be autosomal dominant, autosomal recessive, or X-linked, and each pattern carries different inheritance risks for children and other family members.
